Organic Chemistry Isomerism Questions and Answers and Example - Advanced Level
There are several models in organic chemistry questions. Specially, isomerism questions of organic chemistry are based on selecting exact structures for a given specific chemical formula. In this tutorial, answers for each isomerism questions are explained in detail your understanding. In this type of question pattern, understanding of organic chemistry reactions is a key factor.
Question 1: Identify isomers for C4H10O
A is an aliphatic organic compound. It's molecular formula is C4H10O. This A compound does not hold optical isomerism. Also A does not react with aqueous NaOH solution and Sodium metal.
- Draw all possible structures of A.
- One of A's isomerism can be prepared by using only following reagents. Identify that isomerism and complete the block diagram of conversion.

Answer for Question 1
1. Given data and observations should be deeply analyzed by step by step to make decisions on selection of structures and type of compound. (such as an alkane or an alkene or a carboxylic acid).
- This given organic compound A contains only single oxygen atom. Therefore we can assume A can be an alcohol, an ether, an aldehyde and or a ketone.
- It's mentioned that, that A does not react with Sodium metal or aqueous NaOH solution.
- Alcohols reacts with metallic sodium. Therefore A cannot be an alcohol.
- Aldehydes and ketones reacts with aqueous NaOH solutions (aldol condensation). Only formaldehyde (HCHO) does not show aldol condensation. Now, we can eliminate aldehydes and ketones from the assumption list.
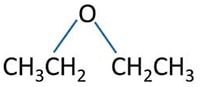
- Now we know, A should be an ether compound.
Let's draw all possible structures of A.
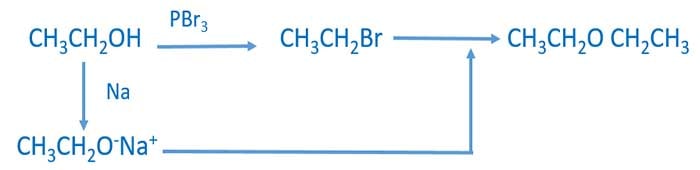
2. Preparing Ether
Ether can be prepared by the reaction of salts of alcohol (like CH3CH2O-) and alkyl halide (like CH3CH2Br).
- We have received ethanol as the only organic compound. Therefore we have to start the conversion from ethanol. We can prepare Sodium acetate (CH3CH2O-Na+) from the reaction between ethanol (C2H5OH) and Sodium.
- Then, Add concentrated HBr acid to C2H5OH to prepare bromoethane (CH3CH2Br) which is an alkyl halide.
- After that, mix both CH3CH2O-Na+ and CH3CH2Br to gey the ethoxyethane as the ether compound.
- Now, we can select our compound as following.
Steps of preparing ethoxyethane from ethanol
Have Questions?