Ammonia (NH3) Molecule Shape, Geometry, Hybridization
Ammonia lewis structure contains three sigma bonds and one lone pair on nitrogen atom. Therefore, there are total of four electrons regions. So, hybridization of center atom, nitrogen is sp3. Because there are four electrons regions, geometry is tetrahedral and shape is trigonal pyramidal.
Ammonia (NH3) lewis structure
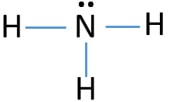
To decide the geometry, shape and hybridization of a molecule, drawing the correct lewis structure is very important.
Number of electron regions in ammonia
Total electron region is taken by the summation of sigma bonds and lone pairs around relevant atom.
Number of electron regions around nitrogen atom in ammonia
According to the lewis structure given above, there are three sigma bonds and one lone pair around the nitrogen atom. Therefore, total number of electron regions around nitrogen atom is four.

Geometry and shapes of molecules according to the number of electron regions
You can decide geometry and shapes of molecules from following table according to the number of electron regions.

Geometry around nitrogen atom in ammonia
Because, number of electron regions around nitrogen atom is four, geometry should be tetrahedral.
Molecular shape of ammonia
Because, there are three sigma bonds and one lone pair around nitrogen atom, molecular shape of ammonia is trigonal pyramidal.

Hybridization of ammonia molecule
Hybridization is also decided by the number of electron regions around an atom. A table is given below for identifying hybridization.

Because number of electron regions around nitrogen atom is four, hybridization of nitrogen atom is sp3
Questions
What are the similar molecules which have similar geometry, shapes and hybridization to ammonia
Phosphine is very much similar to ammonia considering both center atom and outside hydrogen atoms. However, if only center atoms of molecules are considered, nitrogen trichloride, nitrogen trifloride are also has similar geometry, shapes and hybridization to ammonia molecule.